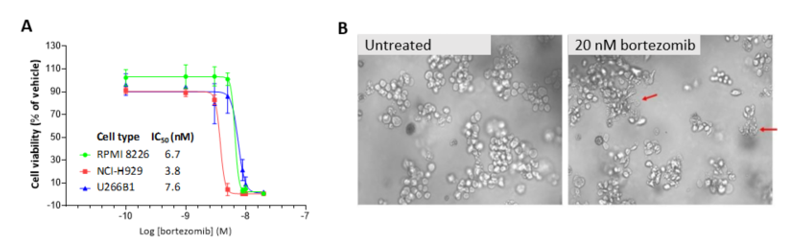
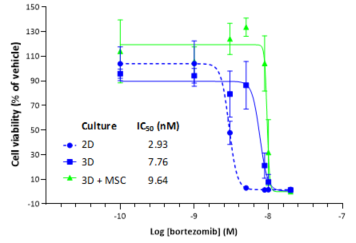
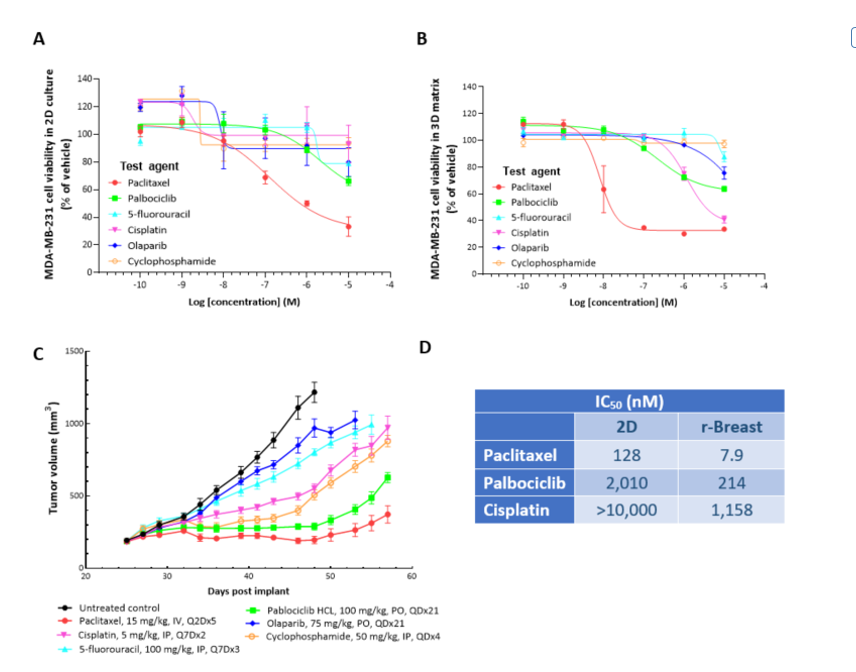
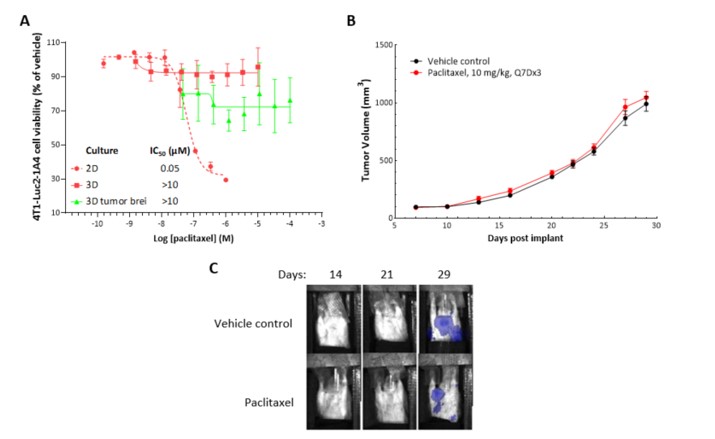
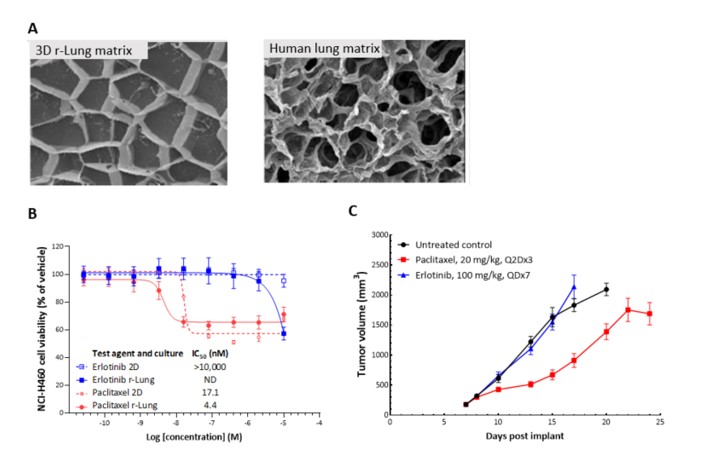
2D in vitro assays do not reflect the tumor architecture and environment found in vivo, resulting in differences in the outcomes and pharmacological potencies of test agents being screened when comparing 2D in vitro versus in vivo models.
To provide a more reflective model of how tumor cells would respond to test agents in vivo, Labcorp and Predictive Oncology®® have co-developed tumor-specific 3D in vitro models that recapitulate tissue physiology and mimic the composition and architecture of both solid tumors and hematological malignancies. This tech spotlight highlights the characteristics of different reconstructed 3D models and their use as tools for in vitro screening of test agents. Here, we have compared responses of tumor cell lines to standard of care (SoC) agents in 2D, 3D and in vivo models.
Highlighted features1:
- The reconstructed extracellular matrix (ECM) allows for co-culture of tumor cells with mesenchymal stem cells (MSCs), fibroblasts, lymphoid, myeloid and CAR T-cells
- Cells can be isolated from the matrix after test agent exposure and analyzed by an appropriate downstream application, such as flow cytometry, RNASeq or in situ imaging; multiple endpoints can be measured in these in vitro assays
- Cytotoxicity assays such as CellTiter-Glo® can be performed in a high-throughput format with the successful development of both 96- and 384-well assay formats
- Multiple types of therapeutic agents can be screened in 3D models, including small molecules, biologics and CAR T-cells
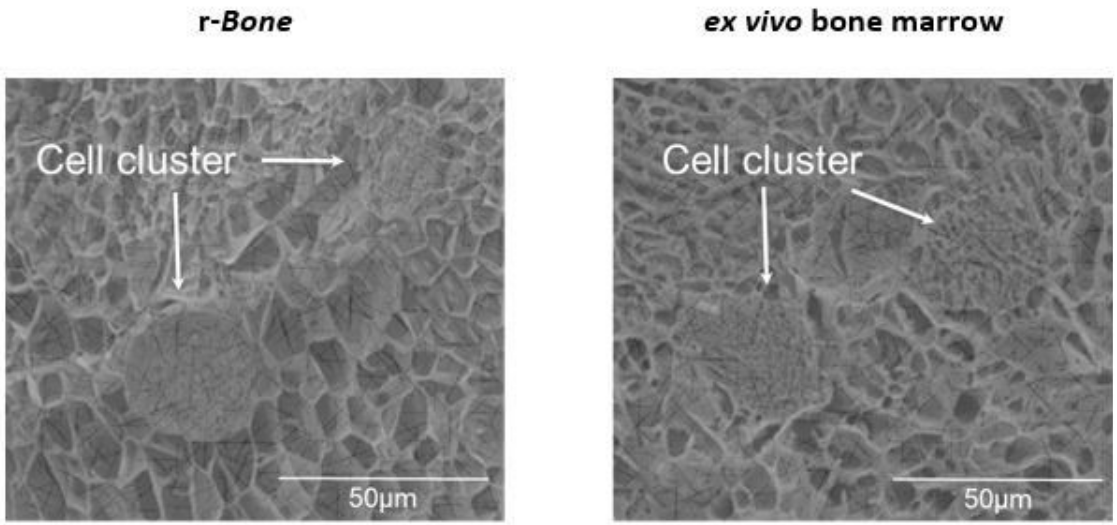
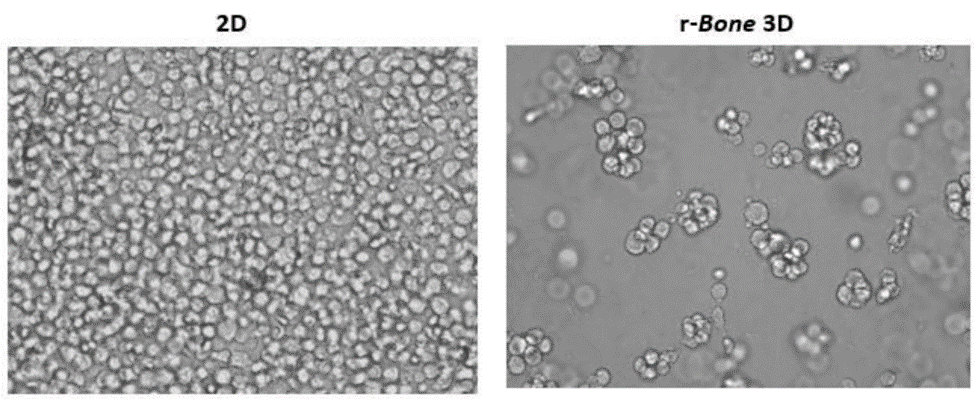
3D reconstructed bone (r-Bone) matrix
The r-Bone matrix is a potential platform for studying multiple hematologic malignancies in vitro as well as for the investigation of solid tumor metastasis, like breast and prostate carcinomas to the bone matrix.