01 May 2017
Author: Erin Trachet, Sr. Scientific Advisor, Oncology | Sr. Manager, Proposal Development
Date: May 2017
It is estimated that in 2017 there will be about 30,000 new cases diagnosed and about 13,000 patient deaths from multiple myeloma. However, it is a relatively uncommon type of cancer with a lifetime risk of 1 in 143 (0.7%). The risk of being diagnosed with multiple myeloma increases as people age, with the majority of people being diagnosed at age 65 or older. Men are slightly more likely to develop multiple myeloma than women and African Americans are twice as likely to develop this type of cancer. However, the greatest risk factor for begin diagnosed with multiple myeloma is already having a plasma cell disease. Patients with monoclonal gammopathy of undetermined significance (MGUS) or solitary plasmacytoma will eventually develop multiple myeloma.
Multiple myeloma is a clonal B cell malignancy characterized by the accumulation of terminally differentiated, antibody-producing plasma cells in the bone marrow. Genetic mutations within the myeloma cells and their interaction with various cytokines and growth factors contribute to the invasiveness of the disease and enhanced drug resistance. Patients are generally asymptomatic until very late state disease. Once disease is detected, localization in the bones, especially the spine, is common. Currently, the gold standard therapy is a combination of doxorubicin and dexamethasone with bortezomib. The addition of bortezomib improves the overall response rate close to 50%.1
A commonly used cell line to evaluate novel therapies for multiple myeloma is MM.1S. MM.1S was derived from a 42 year old African American woman and has been documented to express CD25, CD38, CD52, and CD59. It also expresses the glucocorticoid receptor and is dexamethasone sensitive.2 In an effort to more effectively monitor in vivo disease progression, we transfected the MM.1S line with luciferase (MM.1S-pMMP-LucNeo). With bioluminescence imaging (BLI) we can monitor disseminated disease progression over time and find reproducible in vivo growth in mice (Figures 1 and 2).
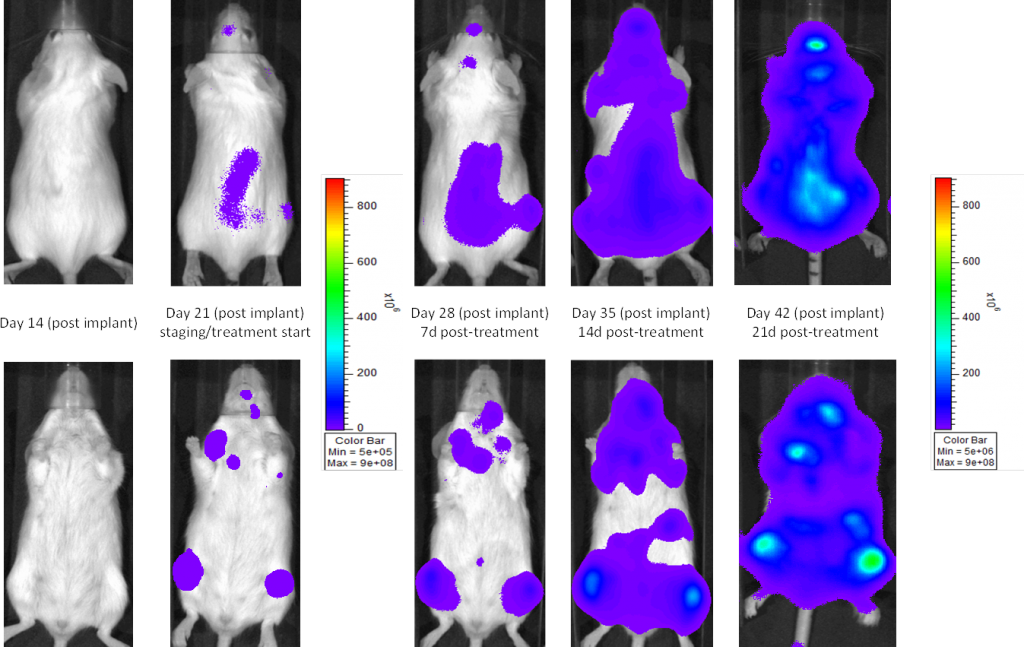
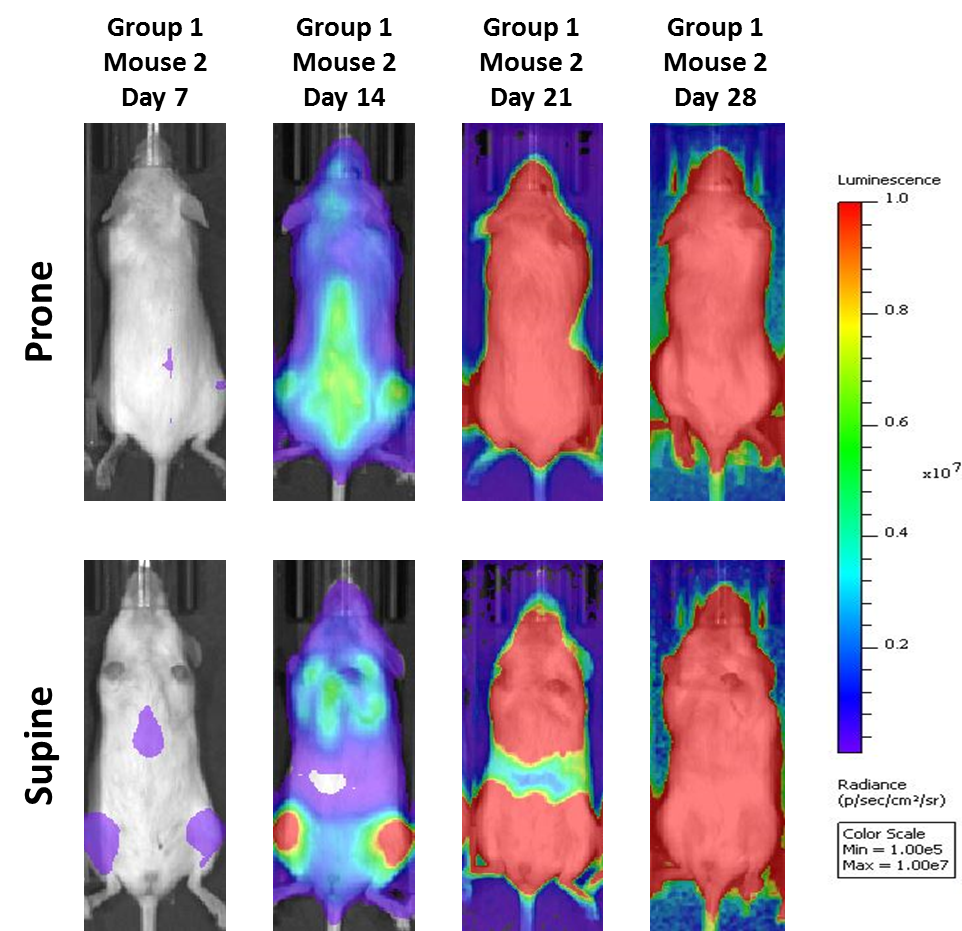
Fig. 2: MM.1S-pMMP-LucNeo human multiple myeloma in NSG mice: Representative Images of Disease Progression – Vehicle Control
Mean Total Tumor Burden BLI Signal
In SCID beige mice, the median tumor volume doubling time is ~2 days (Figure 3), and median time to morbidity/mortality endpoint is 35-45 days. To further optimize the MM.1S-pMMP-LucNeo model, we characterized growth in the NSG mouse strain which shows more aggressive disease onset (staging seven days post implant vs. 15 days in SCID mice). In NSG mice, the tumor volume doubling time is ~1.8 days (Figure 4) and median time to endpoint is ~21 days, roughly half the overall survival time observed in the SCID beige mouse.
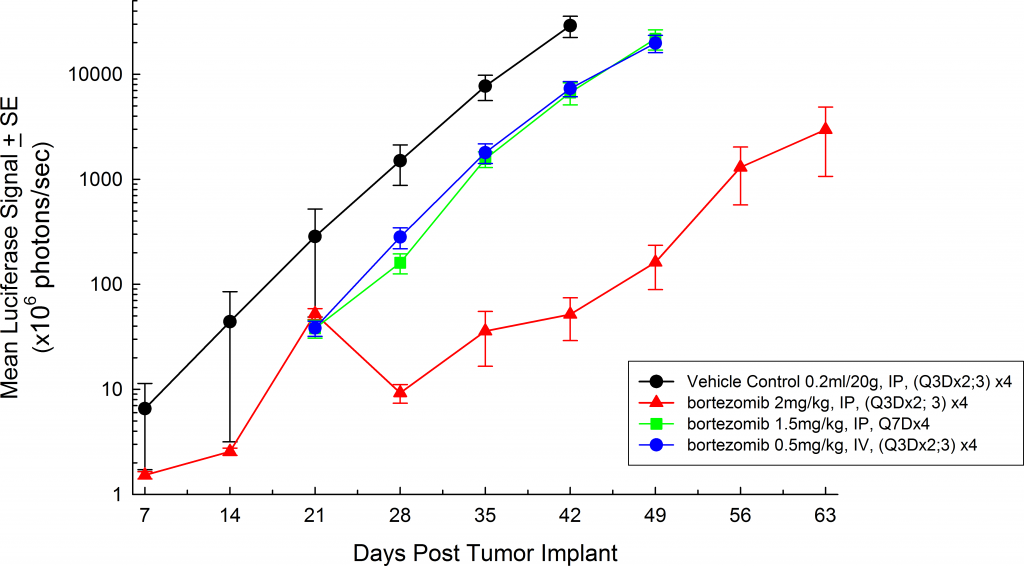
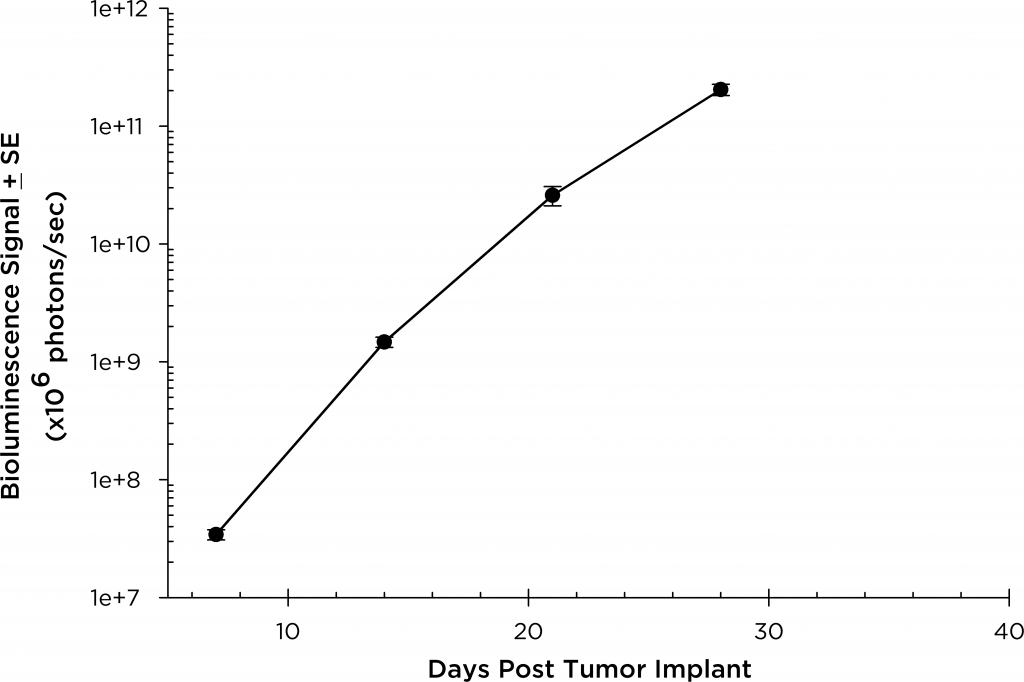
Fig. 3: MM.1S-pMMP-LucNeo human multiple myeloma in SCID Beige mice: Mean Total Tumor Burden BLI Signal
Fig. 4: MM.1S-pMMP-LucNeo human multiple myeloma in NSG mice: Mean Total Tumor Burden BLI Signal
Mean % Body Weight Change
In all mouse strains we’ve investigated, the animals are relatively asymptomatic throughout the lifetime of the study and only begin to present symptoms at late stage disease (Figures 5 and 6); similar to how the disease manifests itself in humans. Common, late stage clinical signs in mice include lethargy and paralysis. BLI on these animals show localization of the signal in the spine, long bones and mandible. Single agent bortezomib (Velcade®) has limited effect on disease progression in this model. This is similar to data seen in the clinic which suggests that response to treatment can be improved when bortezomib is given in combination with other agents. Taking the reproducibility of the model together with translational aspects of progression and response of disease, the MM.1S model provides the platform for evaluating novel single agent treatments as well as drug combinations.
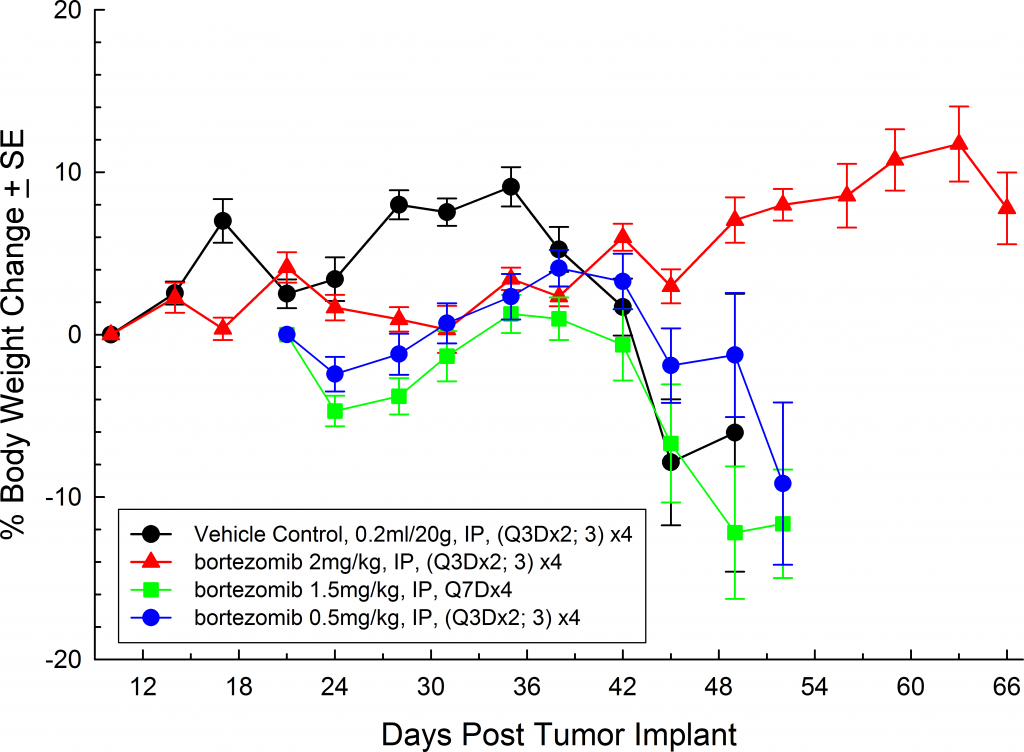
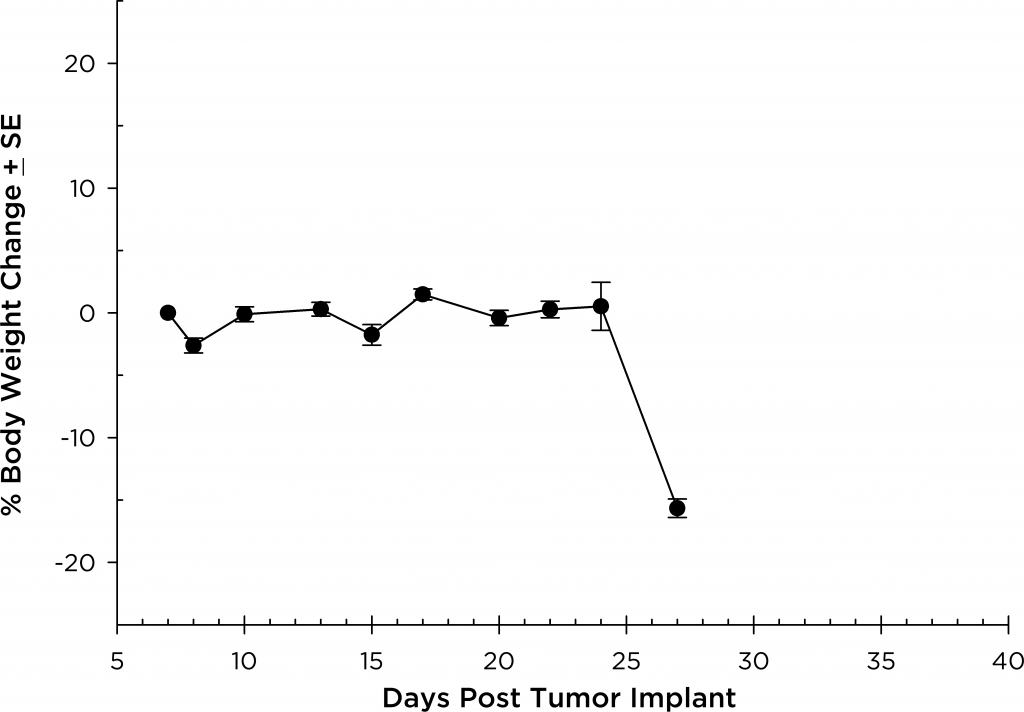
Fig. 5: MM.1S-pMMP-LucNeo human multiple myeloma in SCID Beige mice: Mean % Body Weight ChangeFig. 6: MM.1S-pMMP-LucNeo human multiple myeloma in NSG mice: Mean % Body Weight Change
